
Battery
Measuring powder electrical conductivity with GranuPack
Measure powder electrical conductivity with the GranuPack. A fast, accurate method combining tapped density and conductivity to optimize battery materials and improve electrode performance.
Introduction
The electrical conductivity is the ability of a material to conduct an electric current. For metals in their solid forms, this conductivity is generally high. However, when materials are in powder form, the conductivity is often significantly lower and cannot be predicted from the conductivity of the solid form. Therefore, it is necessary to measure the conductivity in powder form.
The conductivity of a powder is of huge interest in many fields of application. Since it is highly dependent on both the chemical properties of grain surfaces and the arrangement of these grains, a change in conductivity can evidence particle oxidation, moisture uptake, or any change of structure inside the powder. The electric conductivity of powders is particularly useful in the framework of battery manufacturing. Indeed, the electrode needs to have good conductivity to transfer electrons during charge or discharge of the battery, which directly depends on the conductivity of the raw powder material. Therefore, the conductivity of raw material or blend used to produce electrodes needs to be evaluated before the manufacturing process to improve the quality of the electrodes. In addition, the conductivity of a powder is known to increase with packing fraction, since it increases the number of contacts between the particles. This relation is particularly important for electrode production because the materials are generally densified during the manufacturing process, when the powder goes through a calendering process to form the electrode. Nevertheless, the evolution of conductivity with packing fraction is complex to predict. Therefore, the characterization of powder conductivity and how it evolves with packing fraction is of huge interest for battery manufacturing.
In this work, we present a new method to measure powder conductivity coupled with tapped density measurement performed by the GranuPack. This new approach allows evaluating powder conductivity at different packing fractions and how it evolves when the material is densified. Materials of various types, specifically used in electrode production, were tested to highlight the effect of different parameters, such as the proportion of carbon black, the effect of time of mixing, or the level of fibrillation of a powder, on the conductivity. This new method will contribute to the continuous improvement of electrode production, based on powder characterization.
Method
The conductivity of a powder can be measured with the GranuPack by using the conductivity unit and the conductivity measurement cell, as presented in Figure 1. The conductivity cell is used and placed in the GranuPack, replacing the regular one. Before starting a measurement, the two wires of the conductivity cell are plugged into the conductivity unit. This unit is controlled by the software.

Figure 1. (left) GranuPack with the conductivity unit; (right) conductivity cell.
After the regular initialization protocol of the GranuPack, the powder is densified by performing a succession of free falls, called taps. The bulk density and the conductivity are measured and recorded at the same time after each tap, giving access to the evolution of powder conductivity as a function of the number of tap or as a function of tapped density.
Figure 2 presents a sketch of the principle used to measure the powder's electrical conductivity. During the measurement, the powder is in contact with two electrodes, allowing the electric current to pass through. Based on the Ohm's law R=U/I, the resistance R of the powder through which the current passes is computed, knowing the voltage and current intensity measured. The conductivity σ of the powder is then calculated from the equation:
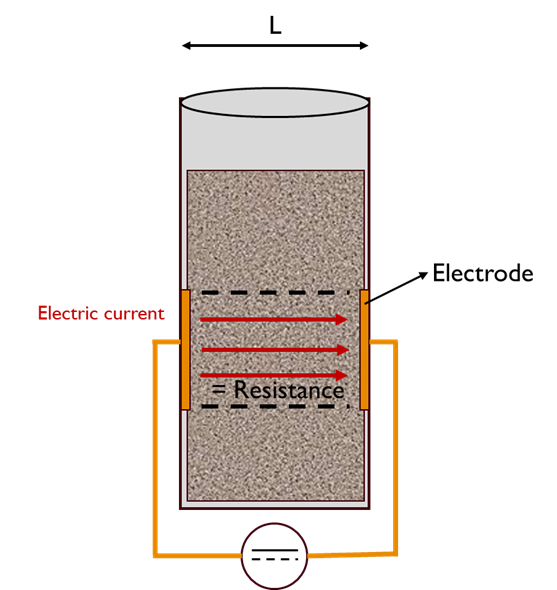
Figure 2. Sketch of the conductivity cell working principle.
Powders Description
- A composite of sulfur (78%) and carbon black (22%)
- Blends of Lithium Iron Phosphate (95%), Carbon Black (3%), and PTFE (2%) at different fibrillation levels
- Blends of Graphite (92%), Carbon Black (5%), and PVDF (3%) mixed for different durations
- A powder of pure Carbon Black
- Model materials: blends of glass beads with carbon black at proportions of 0%, 0.1%, and 1% (by weight)
Results
In Figure 3, the conductivity as a function of the packing fraction η=ρ_bulk/ρ_true is presented for the various powders. Significant differences can be seen between the curves of the different materials and also between the different batches of the same kind of blend. Depending on their composition, the conductivity can be low or very high, with several orders of magnitude of difference. Obviously, the powders containing a large proportion of graphite or carbon black have globally a larger conductivity than powders containing principally insulator materials such as LFP or sulfur. In addition, for each powder, the conductivity increases with the packing fraction. This can be explained by the reorganization of the grains during densification. Therefore, there are fewer voids and more contact between the grains, allowing a better percolation of the electric charges.
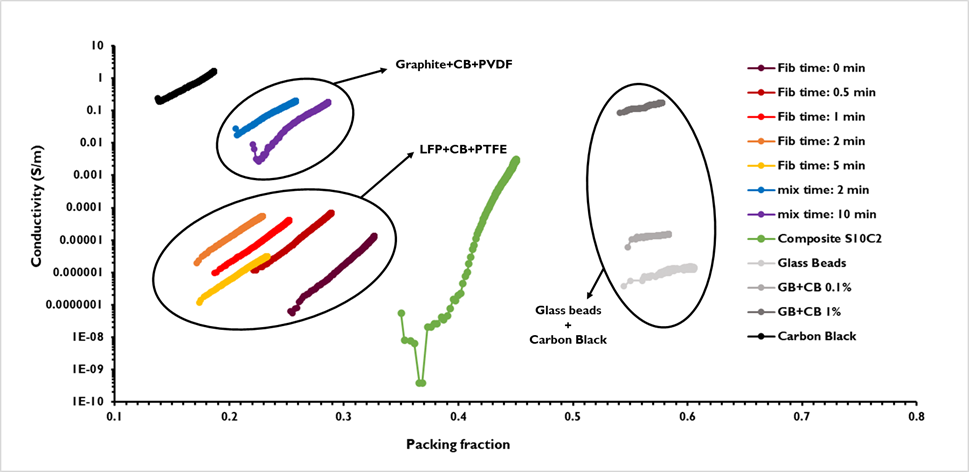
Figure 3. Conductivity as a function of the packing fraction for different tested powders.
This variation highly depends on the powder. Indeed, it can vary by one order of magnitude, such as the pure Carbon Black, or by several orders of magnitude, such as the composite powder of sulfur and carbon, which varies by 6 orders of magnitude between its low and a high packing fraction.
The curves of conductivity versus packing fraction are of huge interest for electrode production since it allows to compare different powder formulations to select the one with the best conductivity to improve the electrode. Furthermore, the evolution of this conductivity according to the densification state of the powder material can be evaluated, giving a better understanding of the dependency of conductivity with material density. This allows a better estimation of the conductivity evolution during material processing, such as after calendaring during electrode production, for which the powder material is highly densified to optimize the contact between the particles and the packing.
Conclusion
The electric conductivity of a powder is an important aspect to consider in many applications. More particularly, it is a key parameter for battery electrode production. Since the electrodes need good conductivity to transfer the electric charges, it is essential to have an adequate electric conductivity of the raw materials in powder form. For this reason, it is important to measure the powder conductivity prior to the manufacturing process to screen different materials to choose the best candidate, adapt the formulation with conductive additive and evaluate how this conductivity evolves with packing fraction to reach the optimal conductive properties for the electrode after calendering.
With the GranuPack, the new conductivity unit with the dedicated measurement cell, the conductivity of a powder can be measured in a fast and automated way in combination with tapped density. The accuracy and repeatability offered by the protocol allow easy powder differentiation and classification according to conductivity and packing fraction. The evolution of conductivity with packing fraction or bulk density can be investigated for a deeper analysis. All these possibilities allow to improve powder formulation, and processing for a better electrode performance.
FAQ – Measuring Powder Electrical Conductivity with GranuPack
What does the GranuPack measure in this application note?
The GranuPack measures the electrical conductivity of powders as a function of packing fraction, simultaneously with tapped density.
Why is powder electrical conductivity important?
Powder conductivity strongly influences electron transport and cannot be predicted from the solid material, especially for battery electrode applications.
How is electrical conductivity measured with the GranuPack?
A dedicated conductivity cell measures electrical resistance during powder densification by tapping, allowing conductivity to be calculated using Ohm’s law.
Why does conductivity depend on packing fraction?
As packing fraction increases, particle contacts become more numerous, improving electrical percolation and increasing conductivity.
Which applications benefit most from this method?
The method is particularly useful for battery materials, helping optimize powder formulation and predict conductivity evolution during electrode processing.
