Papers
Understanding Flow and Packing Properties of Common Nutraceutical Powders
This study demonstrates how advanced powder characterization of common nutraceutical ingredients reveals critical differences in flowability and packing behavior, helping optimize formulation, scale‑up, and reliable manufacturing using GranuDrum and GranuPack tools.
The growth of the nutraceuticals market
Nutraceuticals are biologically active food products that play a role in human health. They are differentiated from regular food products by their nutritional benefits, as well as their therapeutic effects. Therefore, nutraceuticals are classified between food and pharmaceutical products. Nutraceuticals can be used as dietary supplements to supplement the diet, as functional food that provides a physiological benefit, or helps to prevent or control diseases. The demand for nutraceutical products is growing fast, and the global nutraceutical market is expected to reach 675 billion dollars by 2027.
The importance of powder characterization for nutraceuticals production
Nutraceuticals are produced in different dosage forms (tablets, capsules, bulk powder, bags). Each of these dosage forms are associated with processability constraints. The production of tablets requires consistent filling of the die to guarantee low variability of the tablets mass. Moreover, the tablet must have good mechanical properties to prevent breakage during handling. Capsules and bags filling may suffer from lack of flowability, inducing difficulties in the filling process leading to under filling. The processing of the bulk powder can also lead to size segregation that will create variability in the final product.
Less constraint by regulations compared to the production of pharmaceuticals products, the production of nutraceuticals still requires a proper knowledge on powder behavior in order to develop formulations with optimal processability. Furthermore, the rapid growth of the nutraceuticals market will require a scale up of production that will highlight a lack of processability.
Therefore, a relevant powder characterization is essential for nutraceuticals production to guarantee good product quality and reliable processes. In the following, we will see how state-of-the-art powder characterization methods can provide useful information on powder properties to help better understand the material and its processability.
Results
Materials
In this study, we have selected five very common nutraceutical products available on the market: L-Glutamin, Vitamin C, Magnesium, Maltodextrine and Whey protein.
Cohesion and flowability
Powder flowability has been evaluated using the GranuDrum (Granutools, Belgium) rotating drum method. The GranuDrum is a state-the-art method to evaluate dynamic flowability and cohesiveness of powders. The five nutraceutical powders have been tested for rotating speeds ranging from 2 to 60 rpm in order to cover a wide range of stress state and highlight non-Newtonian like behaviors.
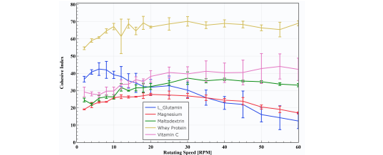
The Cohesive Index (CI), a metric measuring the strength of cohesion i.e. attractive forces acting between the particles, is used to classify the powders cohesiveness. Figure 1 presents the Cohesive Index obtained at different rotating speeds for the nutraceutical powders.
The Whey protein exhibit a significantly higher Cohesive Index than the other powders, denoting a highly cohesive behavior associated with a lack of flowability. Maltodextrin and Vitamin C have a CI between 25 and 30 at low rotating speeds, but see an increase in CI for increasing rotating speeds and thus exhibit a shear-thickening behavior (flowability decreases with increasing stress applied). Magnesium and L_Glutamin powders show the opposite behavior with a decrease of Cohesive Index with speed. Therefore, these two powders are expected to show their flowability improve at increasing process speed. The Whey protein powder demonstrates a Cohesive Index above 60, indicating a high cohesiveness associated with a poor flowability. Processing this powder is expected to be very challenging.
Figure 1: Cohesive Index versus drum rotating speed
Packing behavior
The tapped density results obtained with the GranuPack are presented on figure 2 and principal metrics extracted from the curves are presented in table 1. The Magnesium powder shows the lowest Hausner Ratio (Hr), denoting the best flowability among the testing powder. This is in accordance with the Cohesive index results obtained with the GranuDrum at low rotating speed. The Vitamin C, Whey protein and L_glutamin powders have a similar Hr between 1.36 and 1.39. However, these three materials demonstrated very different flowability in the GranuDrum, with the Whey protein having a much lower flowability than the others. Therefore, the Hausner ratio is not able to clearly assess the differences in cohesiveness of these powders. This is due to differences in packing dynamics of the powders. Indeed, the cohesiveness of the Whey protein powder comes from strong cohesive forces lying between the particles, that prevent an efficient packing of the powder. This is shown by the mobility index (α), which measures the propensity of the material to pack fast. The lower mobility index observed for the Whey protein powder confirms that the low packing amplitude is due to a slow packing dynamic. This highlights the necessity to obtain the complete packing curve allowing an analysis of the dynamics of packing instead of just the initial and final bulk densities.
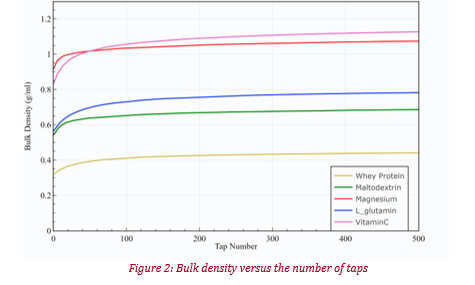
Figure 2: Bulk density versus the number of taps
The permeability of the powders has also been measured with the GranuPack Permeability module. For the sake of conciseness, only the permeability after 500 taps (k500) is reported in table 1. The Whey protein powder has the highest permeability among the powders, denoting a low packing fraction. Indeed, the permeability being the easiness of the air to pass through the material, a higher permeability indicates a lower packing fraction. This confirms the assumption made previously to explain the small difference in Hausner ratio between the Whey protein and L_glutamin.
|
Name |
ρ(0) |
ρ(n) |
Hr |
α |
k500 [m²] |
|
Magnesium |
0,916 |
1,074 |
1,17 |
6,9 |
1,06E-12 |
|
Maltodextrine |
0,541 |
0,686 |
1,27 |
5,5 |
4,20E-12 |
|
VitaminC |
0,830 |
1,127 |
1,36 |
9,1 |
2,19E-12 |
|
Whey |
0,319 |
0,441 |
1,38 |
2,9 |
2,31E-11 |
|
L_glutamin |
0,564 |
0,782 |
1,39 |
5,4 |
7,92E-13 |
Table 1: GranuPack tapped density analysis results
Conclusion
This study highlights the wide diversity of behaviors exhibited by nutraceutical powders in terms of flowability and packing properties. The selected materials show significant differences in cohesiveness, shear response, packing dynamics and permeability, demonstrating how essential a comprehensive powder characterization is for understanding and predicting their processability.
The GranuDrum measurements provided detailed insights into the dynamic flow behavior of each powder, revealing strong contrasts between materials such as Magnesium, showing good flowability, and Whey protein, which exhibited highly cohesive and challenging flow characteristics. Meanwhile, the GranuPack results emphasized the importance of analyzing the full packing curve rather than relying solely on simplified metrics such as the Hausner ratio, which may overlook critical differences in cohesion and packing kinetics.
These findings clearly demonstrate that efficient manufacturing of nutraceutical dosage forms (tablets, capsules, sachets or bulk powders) requires a deep understanding of powder behavior at each processing stage. As the nutraceutical market continues to expand rapidly, advanced characterization tools such as the GranuDrum and GranuPack provide valuable support for optimizing formulations, preventing process limitations, and ensuring robust, scalable production.
FAQ – Powder Characterization in Nutraceutical Processing
Why can nutraceutical powders show poor processability despite similar bulk density?
Bulk density captures only initial and final packing states and does not reflect cohesion, shear sensitivity, or packing dynamics that strongly influence process behavior.
What information does dynamic flowability testing provide compared to static tests?
Dynamic testing reveals how powder flowability and cohesiveness evolve under applied stress and speed, capturing shear‑thickening or shear‑thinning behaviors.
How does packing kinetics affect industrial nutraceutical operations?
Slow or incomplete packing kinetics can lead to inconsistent filling, mass variability, and reduced process reliability in capsules, tablets, and sachets.
Why is powder permeability relevant for nutraceutical formulations?
Permeability reflects packing structure and air evacuation, influencing filling efficiency, compression behavior, and stability during handling.
How can advanced powder characterization support production scale‑up?
By identifying stress‑dependent flow and packing mechanisms, advanced characterization helps anticipate process limitations and reduce scale‑up risks.